Projects
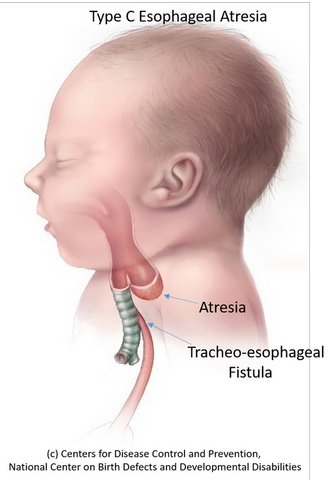
During embryonic development, esophagus and trachea develop from the anterior foregut (AF) tube. Disruption in the separation into two distinct tubes results in foregut malformations such as esophageal atresia and trachea-esophageal fistula (EA/TEF) affecting 1 in 3,500 births. The only treatment available for children born with these malformations involves surgically connecting the deformed esophagus with the stomach with life-long complications.
The mechanisms underlying the embryonic and fetal development of EA/TEF are poorly understood. Much work has focused on understanding the differentiation of the respiratory and digestive system from the AF tube but many questions about the process of compartmentalization remain unanswered. Most of our knowledge on foregut bifurcation, and subsequent esophagus development is based on genetic mouse models which revealed key genes, molecular pathways and signaling molecules that regulate foregut separation andcontinue to play key roles in esophagus development and homeostasis.
Human pluripotent stem cells (hPSCs) provide an efficient system to model and understand human organ development based on mimicking embryonic developmental stages to generate cell and tissue types originating from all 3 germ layers. Our Lab focuses on generation of mature esophageal and respiratory epithelium using induced pluripotent stem cells to better understand the developmental pathways involved in the normal development of the esophagus and trachea.
Our ultimate goal is to use iPSC-derived esophageal, endothelial and mesenchymal progenitors to generate a 3D patient-derived neo-esophagus, to be used as an esophageal replacement in children with long-gap EA without the need of immunosuppression.
LinksParents and Patients Support Groups
The Federation of Esophageal Atresia and Tracheo-Esophageal Fistula Support Groups (EAT)
Association Québécoise de l'atrésie de l'oesophage
External Resources
https://www.cdc.gov/ncbddd/birthdefects/esophagealatresia.html
International Network on Esophageal Atresia
U.S. National Library of Medicine - Esophageal Atresia and Tracheoesophageal Fistula
Database of Genotypes and Phenotypes (dbGAP, NIH)
Esophageal Atresia/Tracheo-Esophageal Fistula (Online Mendelian Inheritance in Man)